Usually, we don’t notice a gene until a mutation changes it. This is true for the vestibular system of Leopard Geckos. Around 2006 certain leopard geckos began to display equilibrium deficiencies, often (but not always) associated with the Enigma morph. These disoriented geckos were diagnosed with a disorder dubbed Enigma Syndrome.
What is ES?
Enigma Syndrome (ES) is a neurological disorder that affects the balance and cognition of leopard geckos. The severity of this genetic mutation varies from relatively mild symptoms, such as: ‘star gazing’, head tilting, and occasional circling to debilitating effects such as: seizures, ‘death rolls’, and incessant circling in place (similar to diagnostic symptoms in autistic individuals). Affected individuals with the same mutation will manifest different degrees of severity of this disorder, because of environmental factors and their individual genotype.
Environmental stressors such as bunking with an aggressive cage-mate, being vacuumed up by a distracted breeder, or just the normal rigors of shipping can be enough trauma to activate this syndrome. Enigma Syndrome is a misnomer, as all morphs are susceptible to this disorder; however, it is more prevalent in the Enigma morph. Balance disorders seen in non-enigma morphs may, in fact, be one and the same disorder.
How is it inherited?
I will preface the following declaration by saying this is my opinion, based on scientific investigation and collaboration with experienced breeders who have produced thousands of enigmas.
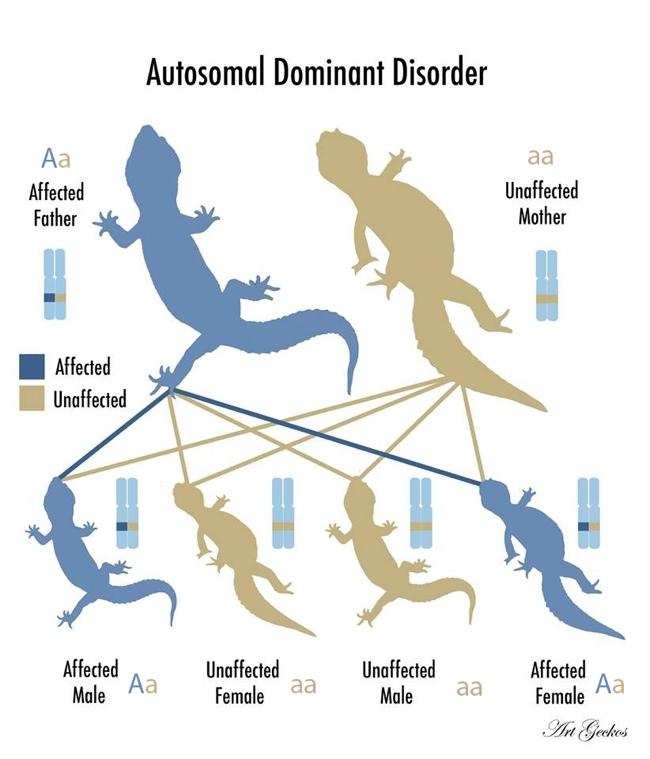
There are 19 pairs of chromosomes in a normal leopard gecko cell. Each animal has two copies of each gene, called alleles. Just one copy of the dominant allele is sufficient to manifest symptoms. Enigma Syndrome is inherited in an autosomal dominant pattern. Those affected are either heterozygous or homozygous dominant. In other words, allele A causes the disorder, so those who are AA or Aa are affected. Homozygous recessive animals, aa, are 100% normal, as they do not carry the mutant allele, and cannot pass it onto their offspring.
The majority of leopard geckos that are affected are heterozygous, Aa. This is because, in many cases, the homozygous dominant genotype, AA, is lethal and usually results in the miscarriage of an embryo.
Every affected animal has an affected parent. If one parent is a heterozygote, the babies will either inherit the gene or not [Fig 1.] (50% chance of inheritance). Both males and females are equally likely to inherit the mutation and be affected. This is because leopard geckos do not have sex chromosomes, so these genes are located on autosomes (non-sex chromosomes).
So, what’s really going on within these equilibrium deficient reptiles?
There are many different types of mutations, but one possible explanation for a disruption in this ES gene is a loss of function mutation, where ES affects brain cells responsible for coordinated movement. Because one copy of the gene is nonfunctional, it produces proteins that are misfolded, such that when they interact with normal proteins they clump up and lead to a toxic disruption of structural organization. Cells can no longer function normally, cell death occurs, and parts of the brain are destroyed. This is very similar to Huntington’s disease in humans, which is an ‘expanding triplet repeats’ mutation. This means the normal gene is packed full of additional CAG repeats, leading to the production of a non-function protein. Again, this is just one of hundreds of possible explanations; until further investigation is done, this will remain an “enigma”.
Often, ES is a late onset neuro-degenerative disease. So you can outbreed a normal enigma, only to later find out they are affected, and the mutant gene was passed to the offspring. Moral of the story: Only breed unrelated, ‘unaffected’ individuals, and outbreed to a different population of genes.
What led to this conclusion?
There is a great deal of research yet to be done, as detailed elsewhere in this article. Consequently, we are at a stage where a hypothesis has been made and needs further testing. I am working with a coalition of breeders whose website is under construction at Gecko Genetics.
What we are fairly sure of, based on breeding and careful record-keeping, is the following:
The disorder behaves as if it is linked to the enigma gene, meaning the two genes are likely located very close together on the same chromosome, such that when crossing over occurs, it’s unlikely the two genes will be separated onto different chromatids during meiosis. This would explain the strong correlation between the enigma morph and the ES gene.
There are, however, unaffected enigmas. Only symptom-free enigmas are bred here at Art Geckos. We outbred an ‘unaffected’ (no signs of ES by 1yr.) female enigma to a different population of genes, and produced 12 healthy animals, 7 of which are enigmas. The mother remains unaffected and shows no symptoms of ES. All of her children behave normally, and are so far unaffected. We also outbred an ‘unaffected’ male enigma, and produced 6 enigmas; a couple of these babies do display signs of ES, so the father must be a carrier even though he hasn’t developed symptoms yet (these animals are now retired as pets). This supports the idea that this is a dominant disorder.
How to solve this problem?
For those animals already affected, there may be some rational solutions available, such as clinical trials of anti-epileptics. The ultimate goal is to avoid producing abnormal animals, which means we must identify carriers of the ES gene. Of course, the only way to verify that an individual is not a carrier, and truly unaffected, is by whole genome sequencing. Finished in 2003, ‘The Human Genome Project’ took 13 years and over one billion dollars to complete. Today this can be achieved in an afternoon for a few hundred dollars. The times are a’ changin’ folks, and this same biotechnology can be utilized to screen each potential breeder for a mutation and determine their risk factors. This way every breeding project will begin with guaranteed normal foundation stock. Additionally, we can track other inherited traits and determine the genetic antecedents. This would eliminate the need for test breeding animals to determine their genotypes and provide precise information about an individual’s lineage.
To sequence an animal’s genome, a sample of the animal’s DNA is collected (in the form of shed, blood, or embryonic fluid, etc.) and sent to an appropriate certified lab. DNA is extracted from the cells and hundreds of copies are made so there is enough DNA to analyze. The genetic material is pipetted onto a DNA chip, incubated (so the DNA attaches to the chip), and the chip is read by a computer, revealing the genes at play. The first step is to sequence the genome of a healthy wild-type leopard gecko. This will provide the normal genetic markers against which other morphs can be compared. Any mutation (morph) can be sequenced using this method.
It is my opinion, based on research and results, ES is an autosomal dominant disorder. It is not autosomal recessive, and it is not an X-linked disorder (as leopard geckos don’t have sex chromosomes)
To pinpoint the exact problem, three things need to happen:
- Macro Pathology – to determine the overall physical damage to the animal
- Micro Pathology – to understand the cellular components at fault
- Genome Sequencing – to genotype and identify the mutation
The first two items will require necropsy of multiple affected animals, while the third item is a straightforward series of laboratory tests. To achieve these goals “The Coalition of Health & Genetics for Leopard Geckos” has recently been established. This collaboration of breeder brains, working with geneticists, should lead to a consensus description of what exactly is going on, so we can eliminate the problem. Not only is it technologically feasible, it is an ethical necessity to produce healthy, well-balanced leopard geckos.






How do I sign on as an interested party to the Gecko Genetics project?
For serious.
Hello, you don’t need to publish this if you think it is way off base. I was thinking since I first heard of the circling and balance thing with geckos about the same thing in dogs that are used just as breeders and not also pets with free roam of the house and a yard; just left in a pen 24/7. After too long a period of no “roaming” they start displaying all the symptoms you are describing. Once started it is too late to reverse (i.e. you could put the dog in the middle of a field and it will still turn in circles in one spot). I always tell people that reptiles “got brains; they just are tiny ones”. Could it be mental or emotional? I have always felt those “racks” are neat in that they house a large amount of critters in a small space but is it really good for the WHOLE critter. If you google SKINK ON HAMSTER WHEEL you will see lots of different lizards you can click on that are running on their hamster wheel…there is even a tegue on a treadmill running his legs off. Some run on the inside of the wheel and some run on the outside of the wheel but I bet it makes them feel great. Both Mentally and physically. When a critter that is used to roaming freely gets contained in a very small box with only a trip to the water dish, a few steps to the hide box and few steps to food there is a lot of time left in their day to wonder what to do next. In zoos the big cats (lions, tigers, etc) were not doing well till someone figured out that they are hunters and feeding them in a bowel like a housecat just didn’t cut it for them. They now put the food on a track and it is pulled across the pen really fast and the cat has to go catch it or wait till next time to eat…just like in the wild. It seemed to satisfy whatever they were missing and they started living longer, breeding better, and actually started to thrive. They also had some circling behaviors in really small zoos with really small pens. I have seen this behavior in many different critters that don’t get enough exercise so I was wondering about either mini treadmills for the racks or they now have “hamster wheels” shaped like a saucer that takes up less height and room but gives lots of time to “in the critters little brain” to run as long and “far” as they want. Just a thought. Everyone or everything needs a road trip now and then.
over 200 babies from various strains of stripe , eclipse , giant and raptor bred in captivity with no enigma genetics ever introduced into my projects .
to date, not a single case of circling, star gazing or any enigma syndrome issues.
in my opinion it’s highly unethical the way these were sold , marketed and disseminated purely in a selfish manner when enigmas first came out,
as a trait that has now infiltrated every morph phase there is , I feel happier knowing that my geckos do not – and will not be carrying enigma genes .
I refuse to advocate breeding broken animals
I was criticized for speaking out about this issue years ago when breeders were selling anything they could cross with the enigma gene,
Told that I was ‘taking food off their tables’… but if you need to lower the standards you should be upholding to make quick money then msybe you should get a day job
Have there been any studies involving calcium deficiency in geckos with enigma syndrome? I’ve had some with moderate to severe that have seemed to do better if every meal was dusted with calcium and/or liquid calcium was in their drinking water. I adopted one out as a “special needs” gecko to a man that happened to work for a vet. They did a blood test and found a lack of calcium in the blood. They injected her with calcium to up levels and she’s now fed a regular diet of bugs that have been injected with liquid calcium weekly. Last time I spoke with him, the gecko was symptom free. It’s observational only so i was wondering if there were any actual studies.
It’s a shame that people still insist on breeding Enigma Syndrome Leopard Geckos these days. The lengths people go for flashy patterns and colors…
“Symptom-free” Enigma Leopard Geckos could still be suffering, and nobody really knows it. Nobody knows the scale, nor does anybody know if you can even breed out the negative traits.
People will constantly try to find excuses to breed these broken animals, yet there is nowhere near enough resources for general, correct care of them in the first place. People still don’t give the basic advice for avoiding impaction: observing your pet! Do you really think that people unwilling to watch their pet eat to find the correct substrate (even though a mix of large rocks and sand is what you actually need) will be willing to look for their enigma syndrome acting up?
You can’t even agree on whether UV is needed. “Extra” means “yes you should buy it so your pet has the best life possible”.
You raise a number of interesting points, many of which are addressed in a variety of Gecko Time articles. It is true that there isn’t complete agreement on some issues, but that’s largely because there’s more than one way to do some things.